|
2/10/2024 0 Comments Density of water imperial 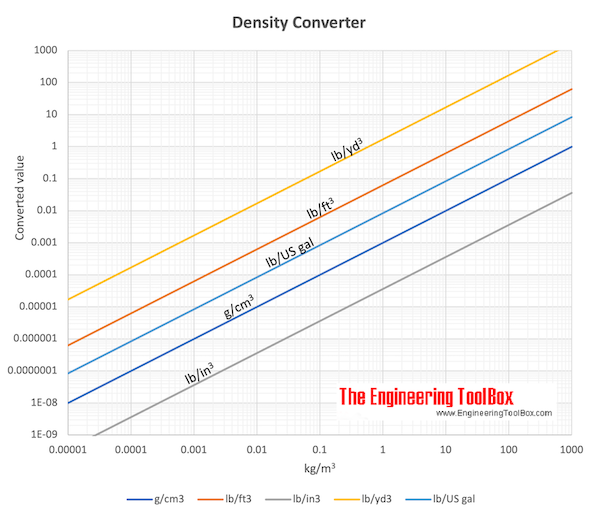
The density of water is the mass of water per unit of volume. Specific Weight for Some common Materials. These electrostatic effect give rise to a shrinkage of the water. What is the density of water in Imperial 1000 kg/m 3 It is common to use the density of water at 4 o C (39 o F) as a reference since water at this point has its highest density of 1000 kg/m 3 or 1.940 slugs/ft 3. Water plays an important role in the world economy as it functions as a solvent for a wide variety of. In this way the field tends to disrupt hydrogen bonded structures in liquid water, and to compress the water molecules surrounding an ion. Water is a liquid at standard ambient temperature and pressure, but it often co-exists on Earth with its solid state, ice, and gaseous state, steam (water vapor). The interaction of the electrostatic field of an ion with water tends to align the dipolar water molecules in the direction of the field. See this excerpt from a study on the volumetric effects due to ion-solvent interaction in aqueous electrolyte solutions: Density 20 C., 1 atm.: 1.205x10-3 g/ml Specific Volume 60 F., 1 atm.: 13.3 cu.ft/lb Thermal Conductivity, Gas 0 C. This calculator will compare the substance density you have entered with the density of pure water and display the result as a specific gravity decimal ratio. 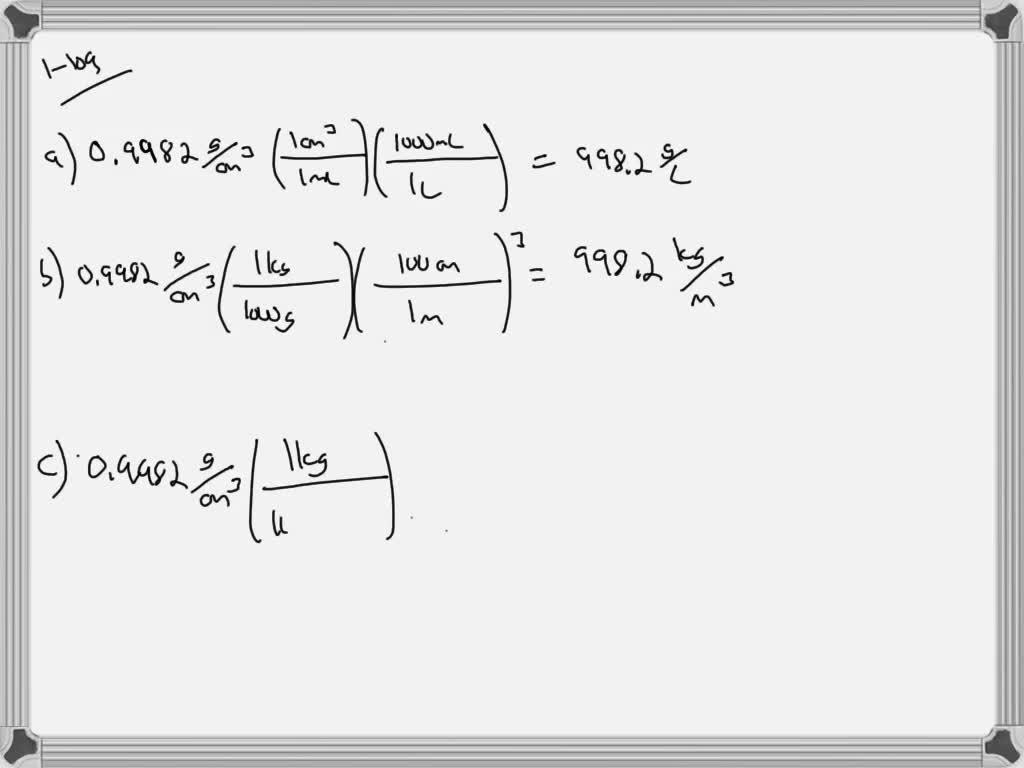
In this more ordered arrangement, the ions effectively fill the voids between the water molecules, and the volume of the water only increases slightly. The positive Hydrogen ends of the water molecules are attracted to the negatively charged Cl ions, and the positive oxygen ends are attracted to the positively charged Na ions. Because of the geometry of water molecules, they are essentially dipoles with “positive and a negative ends”. The density of any substance will change as factors. The Density in Water Density is equal to the Pound per Cubic Inch (pci) multiplied by 0.036127292000084. In imperial measurements, its about 62.4 pounds per cubic foot at 77 degrees Fahrenheit. To convert any H2O measurement to lb/in3, use this formula: H2O lb/in3 x 0.036127292000084. Interestingly, the dissolved salt does not increase the volume of the water by the volume of the added salt, and this is due to the charge of the Na and Cl ions and the H 2O molecules. So, 10 H2O is equal to 0.36127292000084 lb/in3. Specific heat (C) is the amount of heat required to change the temperature of a mass unit of a substance by one degree. 10.022417 pounds assuming the density of water is 1 g/cm 3). An imperial gallon of 160 imperial ounces of water weighs very nearly 10 pounds by design (8.34540449 pounds vs. When salt (sodium chloride or NaCl) dissolves in water, there is a significant increase in mass of the solution due to the relatively higher molecular mass of the dissolved ions Na (22 g/mol) and Cl (35.5g/mol) when compared to water or H 2O (20 g/mol). Online calculator, figures and tables showing specific heat of liquid water at constant volume or constant pressure at temperatures from 0 to 360 C (32-700 F) - SI and Imperial units. A US gallon of 128 US ounces of water weighs about 8 pounds by accident. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
